Challenges in the Disinfection of Ultrasound Probes

Below is a whitepaper featured in Infection Control Today, written by PCI Medical (now CIVCO Medical Solutions).
INTRODUCTION
With over 6,000 customers using more than 18,000 GUS Disinfection Soak Stations™, we at PCI Medical have a unique perspective on the disinfection practices in the ultrasound market. Our Disinfection Specialists speak with our customers several times a year; and as such, we have an inside look as to what Joint Commission inspectors are looking for at healthcare facilities across the country. Moreover, we always have advised our customers to follow published best practice guidelines based on what works best for them, regardless of their selected disinfectant. With that said, let's take a look at the current disinfection challenges that our customers are faced within the ultrasound field.
EVOLUTION OF HIGH-LEVEL DISINFECTANTS
Over the years, we have observed the changing tides in guidelines due to market forces and new disinfectant manufacturers' products. The GUS Disinfection Soak Stations™ for ultrasound probes were first introduced eighteen years ago. At that time, glutaraldehyde was the most commonly used high-level disinfectant (HLD). Endocavity ultrasound probes were soaked in large open bowls/containers or, in some cases, only wiped down with a germicidal wipe. Staff and patients were exposed to toxic vapors and expensive probes were often damaged due to improper handling.
PCI Medical saw the need for an improved disinfection process and pioneered a range of GUS Disinfection Soak Stations that incorporated a vapor control system to eliminate toxic vapors. The GUS systems were designed with custom-made soak and rinse containers that were developed for specific medical instruments. The result was a disinfection system for ultrasound probes that today is the most widely used system in medical facilities across the country. GUS Disinfection Soak Stations provide an additional level of engineering controls that minimize staff exposure by meeting recommended ventilation standards; protect delicate probes from damage during the disinfection process, while using about one-third less HLD than many containers.
As a result of increased attention to engineering control requirements, glutaraldehyde has been replaced for the most part by OPA-based HLDs. When originally introduced, the Safety Data Sheet (SDS) for OPA had minimal safety engineering controls. However, now the SDS for OPA reads virtually the same as that of glutaraldehyde. They both require ten air exchanges per hour, or use with a ductless fume hood (AKA disinfection soak station or vapor control system), or direct venting to the outside.1, 2
Revital®-Ox RESERT® XL-HLD (RESERT), manufactured by STERIS Corporation, is a relatively new FDA-cleared hydrogen peroxide-based HLD intended for use with compatible semi-critical devices, such as flexible endoscopes and ultrasound probes. According to the SDS for RESERT, the engineering controls requirement for room air exchanges when using the product shows a benefit over both glutaraldehyde and OPA.3 Whereas the aldehyde-based HLDs require ten air exchanges per hour, RESERT only requires six in an exam room or ten in a soiled room.
Despite the lower requirements for air exchanges, hydrogen peroxide has an OSHA PEL (Permissible Exposure Level) of 1 ppm and is expected to be used in appropriately designed and operating ventilation systems.4 ORs and most endoscopy reprocessing suites have significantly more than six air exchanges; however, typical exam or reprocessing rooms where ultrasound probes undergo HLD often do not. In addition, heating/air-conditioning systems in these facilities may have both the fresh air supply and return vents located near each other in the ceilings. This design may result in the fresh air taking the shortest route across the ceiling to the return vent leading to inadequately recirculated air within the room. So, even if rooms have six or more air exchanges per hour, chemical vapors can dwell in poorly designed or ventilated spaces and settle within the employee's breathing zone; and therefore, it is important to consider secondary containment risk reduction measures such as a GUS Disinfection Soak Station while using HLD product.
What's the lesson here? All high-level disinfectants, including glutaraldehyde, OPA, and hydrogen peroxide, are designed to kill microorganisms. Despite the use of adequate engineering controls, and following the instructions provided by the chemical manufacturer, some users may be sensitive to the HLD being used, and as a result of these sensitivities, may experience irritation or develop varying levels of allergic response. Often, the reactions are delayed and are not seen until after multiple, repeat exposures well after an HLD has been in use. As such, the safest approach is to implement additional engineering controls and always use a disinfection soak station with vapor control when soaking an instrument in any high-level disinfectant.
THE RINSE CONTROVERSY
Many of our customers have been cited by Joint Commission inspectors for not following the rinsing instructions for OPA or glutaraldehyde. The IFUs for these HLDs require a large volume of rinse water, e.g. two gallons, for three separate rinses (up to six gallons) when rinsing a device.5
The challenge is the sheer volume of water. The IFUs were likely written with the largest instrument in mind, e.g. a colonoscope. Two gallons is what you need to fully immerse a colonoscope. The typical colonoscope is very large, with intricate lumens and ports that present special issues with rinsing. An endovaginal/endorectal ultrasound probe, by comparison, is considerably smaller and is made from smooth rigid plastic with no lumens or ports.
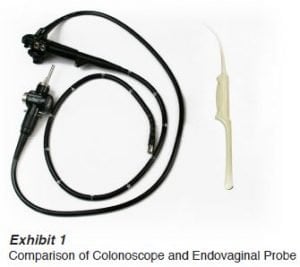
Just how big is a colonoscope compared to a typical endovaginal ultrasound probe? The photo in Exhibit 1 shows a typical colonoscope and endovaginal probe. This colonoscope displaces approximately 1,174 ml compared to only 170 ml for the endovaginal probe. In addition, the surface area of the colonoscope is more than ten times larger than that of the endovaginal probe. Logically, an endovaginal/endorectal ultrasound probe should therefore only need 1/10th the volume of rinse water that a colonoscope requires. That translates into three rinses of only a quart (one liter). This will save valuable counter space, eliminate time wasted filling and emptying large bowls and save 5.5 gallons of water!
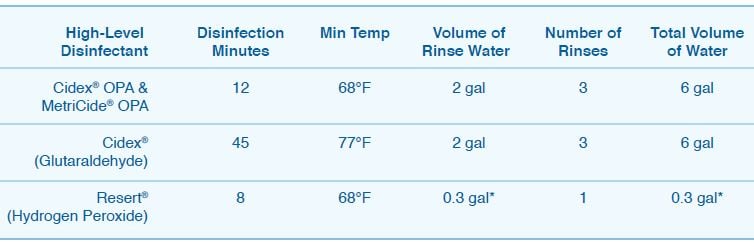
One widely used technique of rinsing endovaginal/endorectal probes after disinfection is to hold it under a faucet of running water for at least a minute. A typical faucet flow rate is approximately three gallons a minute. Although effective in meeting volume requirements, this also doesn't meet the IFU. It is time that the IFUs were changed to reflect the size of the device being rinsed. Some of our customers have recently switched to using RESERT for their high-level disinfection procedures. RESERT is the only ‘ready-to-use' 2% hydrogen peroxide based high-level disinfectant that is FDA-cleared. The following chart shows comparisons between RESERT, CIDEX OPA and Metricide OPA, and CIDEX (glutaraldehyde-based), according to their IFUs when disinfecting and rinsing an endovaginal/rectal probe.
REPROCESSING TIMES
Total cycle time is very important. Not only does RESERT significantly save on rinse water (at least 5.7 gallons per cycle), but it also shaves off 7.75 minutes of reprocessing time when compared to OPA (that's nearly 50% faster). The following is a brief overview of the reprocessing steps based on a simulated disinfection cycle of an endovaginal/probe.
- CIDEX OPA & Metricide OPA require 12 minutes for manual disinfection at 68°F; 2 minutes to fill and empty 3 separate 2-gallon rinse bowls; plus 3 minutes for the 3 separate rinse/holds for a total of 17 minutes.
- Resert requires 8 minutes for manual disinfection at 68°F; 15 seconds to fill and empty the GUS rinse container; plus one minute for rinse/hold, for a total of 9.25 minutes.
In addition, RESERT can be reused for up to 21 days or until such time as the MRC test strip fails, whichever comes first. CIDEX OPA and Metricide OPA have a 14-day reuse period.
MAINTAINING TEMPERATURE
Maintaining disinfectant temperature has also been a 'hot' topic for Joint Commission Inspectors. Both OPA and RESERT require a minimum of 68°F when manually disinfecting. The quick fix for OPA and RESERT is to set the room temperature to 70°F, but this is not always possible. It is even a greater challenge with glutaraldehyde-based HLDs that require a minimum of 77°F. PCI Medical has developed a small warmer designed specifically for the soaking containers used to disinfect endovaginal and endorectal probes.
SOAKING DEPTH
 Just how much of the probe should be
disinfected? In the last six months, we started to hear from our customers that Joint Commission inspectors had
specifically looked to see that the probes were disinfected above the handle. The handle and the area directly above
it are often touched during an exam and could easily be a source for cross contamination if not properly
disinfected. One must refer to the probe manufacturer's instructions. In response to this, PCI developed new soak/rinse containers that
allow the probes to
be disinfected 3.5 above the handle (marked in photo Exhibit 3).
Just how much of the probe should be
disinfected? In the last six months, we started to hear from our customers that Joint Commission inspectors had
specifically looked to see that the probes were disinfected above the handle. The handle and the area directly above
it are often touched during an exam and could easily be a source for cross contamination if not properly
disinfected. One must refer to the probe manufacturer's instructions. In response to this, PCI developed new soak/rinse containers that
allow the probes to
be disinfected 3.5 above the handle (marked in photo Exhibit 3).
CONCLUSION
Ultrasound departments are faced with their own unique issues when it comes to high-level disinfection. Challenges related to safety, rinse, time, temperature and probe disinfection depth are among the most common. Practitioners should weigh in on these factors when they choose their disinfectant. OPA has been on the market since 1999 and has been approved for use by major probe manufacturers. To date, probe manufacturers such as Philips and Siemens have approved RESERT for use with a majority of their probes. STERIS has done extensive compatibility testing and positive user acceptance has grown over the five years it has been on the market. We encourage our customers to check with their probe manufacturer to confirm compatibility and to choose the disinfectant that best meets their needs.
References
- ASP® Cidex® OPA Instructions for Use. Ortho-Phthalaldehyde Solution high level disinfectant for semi-critical medical devices, 20390
- ASP® MSDS Cidex® OPA MSDS-09588-0-001
- Steris SDS Resert XL HLD 4450
- https://www.osha.gov/dts/chemicalsampling/data/CH_246600.html
- ASP® Cidex® OPA Instructions for Use. Ortho-Phthalaldehyde Solution high level disinfectant for semi-critical medical devices, 20390







